CELLIANT® undergoes rigorous testing via a comprehensive and unique set of methods which ensure quality control and efficacy.

When a brand partner works with CELLIANT technology, we understand that they need to be able to stand fully behind its claims.
As such, the CELLIANT team has gone to great lengths to determine a variety of testing methods to confirm both the presence of CELLIANT and its efficacy in providing its stated benefits.
The CELLIANT Testing and Research & Development divisions work with brand partners to determine the best testing protocol for their needs, which can vary based on the product category, application method and other factors.
Regardless of the product or application, brand partners can rest assured that CELLIANT will guide them through the testing protocol most appropriate for their innovation, working to establish the highest level of quality control.
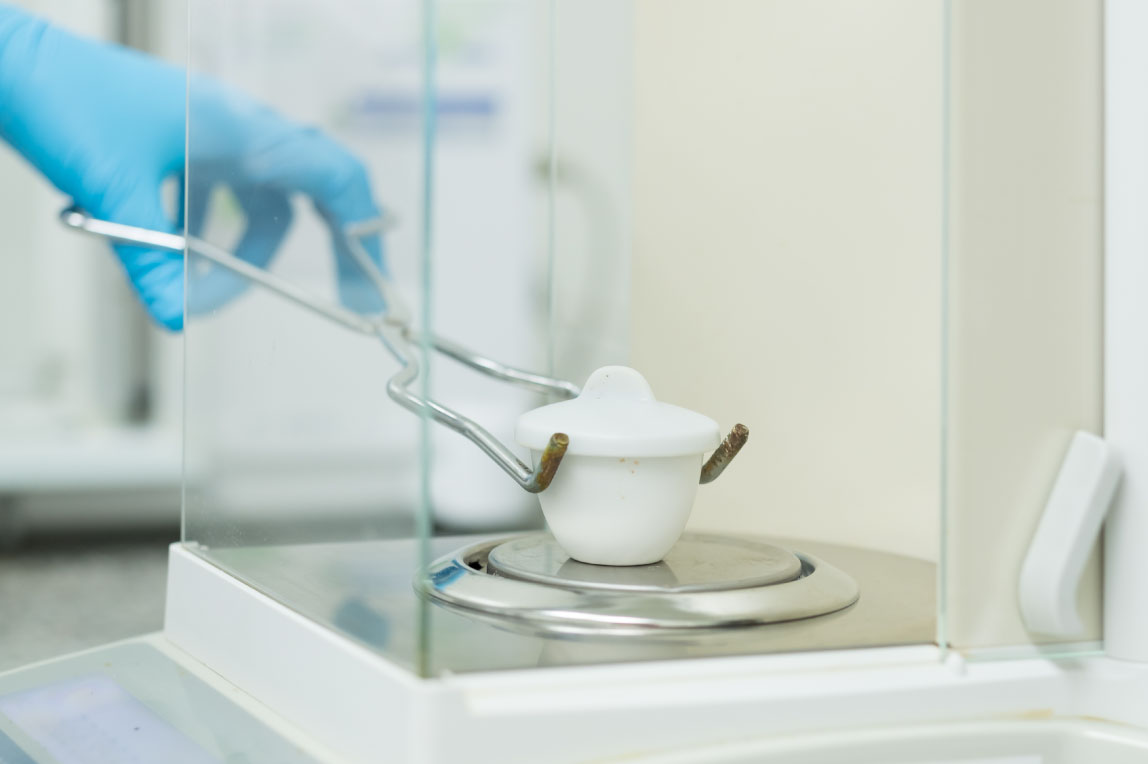
Ash testing is used to prove that CELLIANT minerals are present in our partners’ products in the required amounts.
Sample products are sent to a U.S.-based, independent, 3rd-party lab where they are ignited to burn off the organic materials, and what remains are the CELLIANT minerals.
For CELLIANT-powered products to pass ash testing, the remaining CELLIANT minerals must be present in the prescribed amounts based on our usage and product loading standards.
Emissivity testing is used to prove that CELLIANT-powered products emit infrared at significant levels.
Emissivity testing is done in a U.S-based, independent 3rd‑party lab.
For CELLIANT-powered products to pass emissivity testing, they must emit an average increase of .25 milliwatts per centimeter squared that is generated in the 2.5.‑20 micrometer range when tested at 35°C.

Transcutaneous Oximetry (TcPO2) testing is used to prove that CELLIANT-powered products increase tissue oxygenation, improving local circulation.
TcPO2 testing is conducted on human subjects at our IRB registered lab, and measures the local tissue oxygen released from capillaries.
For CELLIANT-powered products to pass TcPO2 testing, they must demonstrate an average minimum increase of 7%.
X-Ray Fluorescence (XRF) testing is a technique that uses short-wavelength x-rays to elicit energy from a material in a way that shows the characteristics of the atoms present.
The XRF testing is conducted at an independent, 3rd-party laboratory that specializes in using XRF to analyze the elemental composition of a material.
When a CELLIANT-powered fabric is scanned with XRF technology, it confirms the presence of CELLIANT minerals as part of the composition report.

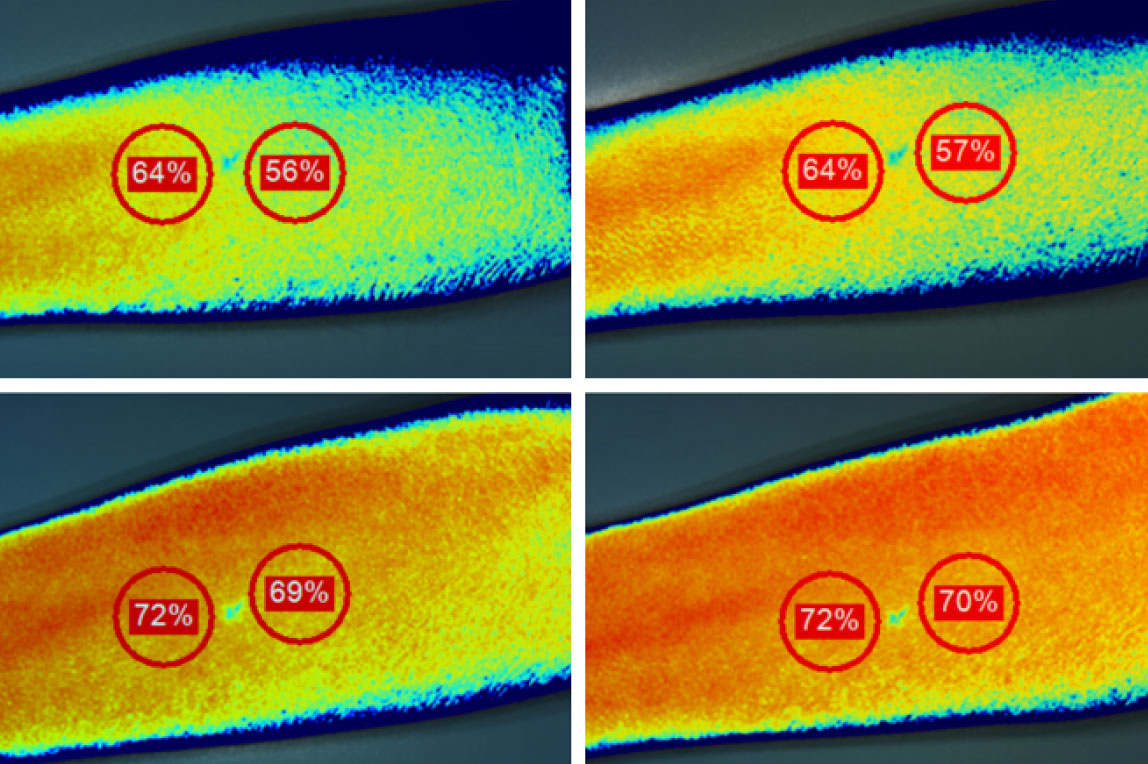
Hyperspectral Imaging (HSI) testing is used to prove an increase in tissue oxygen saturation.
HSI testing is done on human subjects at our IRB registered lab, and uses multiple wavelengths of near-infrared light that penetrates approximately 2-3mm into the superficial tissue to measure relative amounts of deoxygenated hemoglobin in the local area.
For CELLIANT-powered products to pass HSI testing, they must meet a minimum increase of 6% tissue oxygen saturation.
For illustrative purposes, we have prepared a snapshot of our testing procedures for partners curious to learn more about how CELLIANT products are tested. Watch the video to learn more.
For a full-spectrum experience, find related reading about similar topics below.
Products containing CELLIANT undergo testing that confirm that CELLIANT is integrated at the percentage/load required to be effective based on product type and construction. Because of our testing, you can rest assured that any CELLIANT-powered product from one of our authorized brand partners does contain CELLIANT in the proper amount.
TcPO2, or Transcutaneous Oxygen Pressure, is a measure of localized tissue oxygen levels below the skin. Increasing tissue oxygen levels can help to increase local blood flow.
IRB refers to an Institutional Review Board that oversees testing to ensure compliance with standards for research involving human subjects.
To access all of our reports please input your email below.
Contact us at marketing@celliant.com
Thank you for your request. Please download the brochure below.
Sign up for the latest Celliant news and innovations.
REQUEST INFORMATION